
Lips have a significant role in facial aesthetic perception (Yazdanparast et al, 2017).
For this reason, lip enhancement is now one of the most frequently requested treatments in medical aesthetics (Brandt and Cazzaniga, 2008; Kane et al, 2012).
As part of the author's non-surgical facial aesthetics Level 7 postgraduate certification course, numerous lip augmentation procedures were performed on patients who attended clinics. During one clinical examination, one patient who had previously undergone lip dermal filler treatment by another practitioner was assessed. At this appointment, the patient presented with ‘increased lip projection’, extending not only to the vermillion border of her lip, but also beyond this area into the cutaneous ‘white lip’ (Ravichandran and Mann, 2020). According to Sarnoff and Gotkin (2012), the ‘ideal ratio of the upper to lower lip is 1:1.6’. Additionally, they stated that, ‘if a straight line is drawn from the subnasion to the pogonion of the chin, the upper lip should project no more than 3.5 mm anterior to this line, and the lower lip 2.2 mm’. When questioning the patient's treatment history, she reported that she previously had lip fillers placed over a year ago (thought to be Juvederm® Ultra III), to both her vermillion border and body of her upper and lower lip. She went on to say that she felt her lips had changed in appearance over the last year, with the filler ‘migrating upwards’ into her skin, giving them a ‘duck-like’ appearance.
The longevity of dermal fillers
Research suggests that hyaluronic acid lip fillers usually last approximately 6–9 months before the host's immune response degrades them (Kopp et al, 2013).
A study by Allemann and Baumann (2008) supports this theory. However, there is other evidence to suggest that the longevity of these products may be substantially longer (Edwards and Fantasia, 2007), which could explain why the author's patient still had such lip prominence 12 months after treatment.
Kopp et al (2013) suggested that ‘there may be a subset of patients who do not break down hyaluronic acid normally when it is placed in the submuscular plane, leading to un-degraded product migration upward into the dermis’. This notion is supported by a study by Bennett and Taher (2005), who found that the hyaluronic acid filler Restylane was still present in the dermis of a patient's lip 23 months after augmentation treatment. Despite this, there is little evidence to explain how this increase in longevity occurred. However, a study by Narins et al (2008) did find that increased exposure to Restylane (or repeated close proximity injections) can significantly increase the longevity of its products, albeit to a maximum of 18 months. On reflection, the author feels that further research on dermal filler persistence within tissues is required to provide better evidence-based results of filler longevity.
Filler migration
According to Requena et al (2011), the ideal dermal filler should offer good results with a long-lasting effect, as well as being biocompatible and safe with minimal complications or adverse effects and no risk of migration.
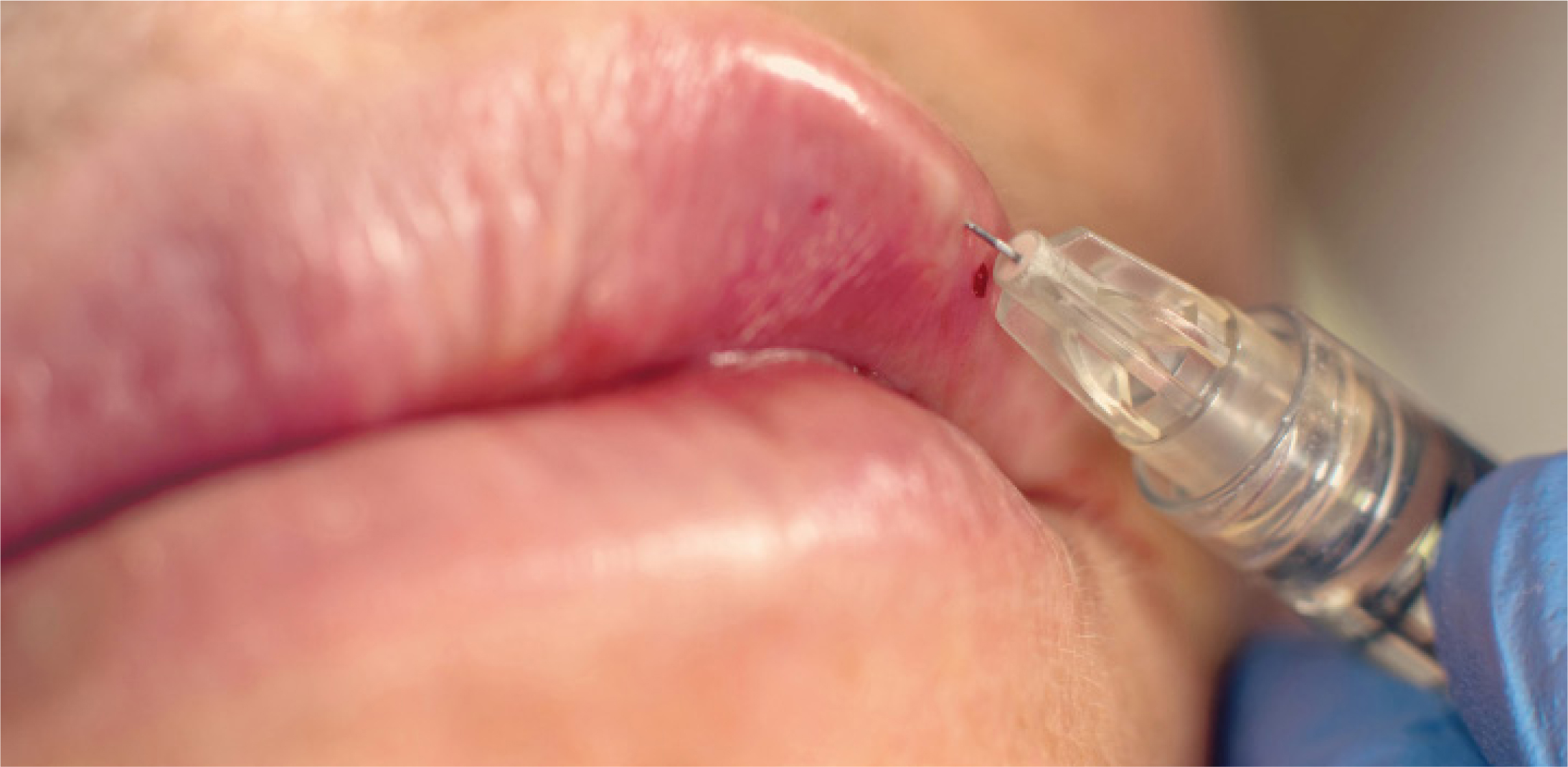 Research shows that silicone fillers are more likely to migrate; however, filler migration after hyaluronic acid injections have also been found
Research shows that silicone fillers are more likely to migrate; however, filler migration after hyaluronic acid injections have also been found
Filler migration is a rare complication of dermal filler treatment (Cox and Adigun, 2011). Research has shown that more permanent fillers (such as silicone) are more likely to migrate, due to their longstanding presence in the body (Broder and Cohen, 2006; Narins and Beer, 2006).
Despite this, there have also been several reported cases of filler migration found from hyaluronic acid-based dermal fillers (Kopp et al, 2014; Chae et al, 2016; Mosleh et al, 2019).
Studies have found that filler migration can occur for several reasons, including poor injection technique (such as incorrect placement of the filler or high-volume/high-pressure injections), increased muscle activity, excessive massaging after filler injection, lymphatic spread and intravascular injection (Jordan and Stoica, 2015). In rare cases, studies have also found that filler can even migrate due to normal skin ageing that leads to changes in facial contour (De Boulle, 2004).
Although many patients may not be aware of subtle changes in filler location over time, others may develop more prominent lesions or lumps away from the initial injection site. These can cause concern for asymptomatic aesthetic reasons, such as the bulky appearance of the white lip seen in the author's patient, but in other cases may be associated with erythema, swelling and even infection (Lafaille and Benedetto, 2010). Migrated filler has also been shown to even mimic granulomatous skin conditions or malignancies of distant organs (Broder and Cohen, 2006; Narins and Beer, 2006).
Foreign body granulomas
Foreign body granulomas are a heavily researched delayed complication of dermal fillers caused by a chronic inflammatory reaction to the products (Lee and Kim, 2015). Studies that have looked at granulomas following lip augmentation procedures have found that these lesions often present as firm nodules. They may be localised at the injection site or away from it, usually located in the submucosa of the maxillary or mandibular sulcus, depending on whether the upper or lower lip was injected (Eversole et al, 2013). This development away from the original injection site could highlight the migration of filler over time.
As a dentist, the author examines the inside of patients' mouths on a daily basis. For this reason, she is often one of the first people to notice any abnormal lump or lesion developing intra-orally. This research has highlighted the importance of enquiring into the possibility of previous dermal filler procedures when presented with a patient who has a new nodular lesion of the lips, oral mucosa or facial region, as this could be included in the differential diagnosis (Eversole et al, 2013).
Lip surface
Extensive research supports the injection of dermal filler in to the submucosal layer of the lips, for several reasons (Tansatit et al, 2014; Lee et al, 2018). It is common knowledge that lips are highly vascular areas (Pinar et al, 2005). For this reason, several studies have suggested that as the main labial arterial branches are most commonly distributed 3 mm below the lip surface, the submucosal plane is deemed safe for injections.
» Filler migration can occur for several reasons, including poor injection technique, increased muscle activity, excessive massaging after filler injection, lymphatic spread and intravascular injection «
Additionally, Bailey et al (2011) found that too superficial a placement of hyaluronic acid filler can potentially result in increased risk of lump and nodule formation, so injecting in the submucosal plane is deemed safer. According to Kim et al (2014), when dermal filler is injected too superficially, particularly in mobile areas like the lips, sometimes filler migration can occur. This theory is further highlighted in a study by Berlin et al (2006), who found that filler migration occurred with superficial injections of calcium hydroxyapatite when injected into the lip. They suggested that while speaking or eating, the orbicularis oris muscle acts as a pump and moves this superficial material around, causing migration and, in some cases, ‘clumps’ material together, which results in nodule formation (Berlin et al, 2006).
However, a study by Blandford et al (2018) found that the majority of hyaluronic acid fillers injected into the vermillion border of the lip resides within the orbicularis oris muscle, rather than in the subcutaneous/submucosal layers. Similarly, a study by Vent et al (2014) agrees with this, finding the presence of both subcutaneous and intramuscular hyaluronic acid filler after injecting into the vermillion border, the body and the philtrum of patients' lips using both needles and cannulas.
Technique
Although there is a lot of research available discussing cannula versus needles as techniques for lip augmentation, there is little information comparing the two techniques for the post-treatment complication of incorrect filler location placement. However, Blandford et al (2018) did find that both techniques seem to regularly produce filler locations within the orbicularis oris muscle, rather than the more superficial layers.
Multiple papers state that the ‘placement of filler into the intramuscular layer of the lip is a contraindication for lip augmentation procedures’ (Lemperle et al, 2006). According to Lemperle et al (2006), if filler is injected into the muscle rather than more superficial layers, constant muscle movement by the orbicularis muscles can compress the injected filler and move it to other perioral areas where it should not be. In addition to this, it is believed that this filler compression can also increase the risk of delayed onset nodules and bumps (Lemperle and Duffy, 2006).
In contrast to this, Vent et al (2014) argued that, for effective lip augmentation, intramuscular injections may sometimes be required. Their theory is supported by Reznik (2019), who states that if a patient has a ‘hyperactive’ lip (i.e. one with increased orbicularis oris muscle activity), intramuscular dermal filler injections may be beneficial, as this could act to reduce the muscles excessive activity and, furthermore, minimise the risk of filler migration.
Regardless of which layer of the lip the dermal filler is injected into, research shows that filler migration can occur from the natural compression of the tissue layers by the orbicularis muscle during normal oral function, particularly in the early post-injection period (Niamtu, 2011). Lemperle et al (2006) described this theory as ‘filler dislocation’, and reported that migration of this nature is most likely to occur in the early hours post procedure. For this reason, it is recommended by some that a patient should avoid any excess oral function for 24 hours following the placement of perioral filler.
High pressure and volume versus low pressure and volume
High-volume and high-pressure dermal filler injections have also been reported to increase the risk of dermal filler migration (Jordan and Stoica, 2015). Beer (2007) suggested that, once injected, ‘dermal filler may travel along planes of least resistance (such as connective tissue planes, superficial musculo-aponeurotic layers, blood vessels or even needle tracts)’ when too much pressure is applied to the syringe. A case report by Goldman (2009) further supports this theory, finding that a patient developed a non-inflammatory, pressure-induced firm nodule on the inside of her upper buccal mucosa following a lip augmentation procedure that involved high doses and high pressures of polyacrylamide gel. This lead to the subsequent migration of this material along tissue planes into the intraoral mucosa. For this reason, low-volume and low-pressure filler lip injections are recommended to minimise filler migration (Beer, 2007).
Excessive massaging
Excessive massaging post injection has also been found to be a potential cause of filler migration (Funt, 2011). The purpose of massage after filler treatment is to mould the dermal filler into an area of volume deficiency so that it appears fuller and without bumps (Jordan and Stoica, 2015). However, too vigorous a massage may move the filler into adjacent areas that were not intended to have filler (Jordan and Stoica, 2015). A study by Ross and Malhotra (2009) supports this theory, describing a 51-year-old female patient who received extensive massaging following nasolabial and cheek augmentation using polyalkylimide 4% filler. Five weeks later, this patient presented with migrated filler to her left lower eyelid. According to Funt (2011), overzealous massaging can result in the filler moving more superficially through the needle tracts created during the procedure. Furthermore, this results in visible material in the skin and can potentially even result in tissue oedema.
Despite this research, many papers still advocate the technique of massaging following lip augmentation procedures (Jacono, 2008; Sarnoff and Gotkin, 2008; Demosthenous, 2017). They report that massaging after injecting helps to attain the desired shape and structure of the lips and, without it, patients are more likely to endure post-treatment lumps and nodules. On reflection, while it is difficult to quantify exactly what is deemed to be ‘too excessive’ a massaging technique, a gentle massage approach could be the answer to stop accidental early filler displacement via this method, while also minimising other post-injection dermal filler complications.
Severe complications
Lymphatic and interarterial spread of dermal filler has also been reported, as complications of dermal filler treatment can result in far more serious issues than those described above. These have been linked with filler migration to soft tissue sites like the eyes, liver, lung, kidney and brain (Lazzeri et al, 2012). However, these are a separate discussion and have been thoroughly reviewed and explored in many dermal filler complication papers (Inoue et al, 2008; Lazzeri et al, 2012; Bravo et al, 2015).
Conclusion
Filler migration is a very real and potential dermal filler complication that can occur from lip augmentation procedures to elsewhere on the face. As a medical aesthetic practitioner, it is important to critically analyse the research available and use it to ensure the safest possible practice is provided for patients, while minimising all potential risks. When performing dermal filler procedures in practice, the author recommends informing patients of the potential risks of filler migration associated with treatment, as well as how these risks will be minimised.
Key points
- Most lip hyaluronic acid fillers report longevity of between 6 to 9 months before the host's immune response degrades them. However, there is evidence to suggest that the longevity of these products may be substantially longer
- Although the longstanding presence of hyaluronic acid in the body could be linked to filler migration over time, further research regarding dermal filler persistence within tissues is required to provide better evidence-based results
- It is believed the ideal dermal filler should offer ‘good aesthetic results and have a long-lasting effect. It should also be safe, biocompatible, and stable at the implantation site, with minimal complications and no risk of migration’
- Studies have found that filler migration can occur for several reasons, ranging from poor injection technique (such as incorrect placement of the filler or high-volume/high-pressure injections) to increased muscle activity, excessive massaging after filler injection, lymphatic spread and intravascular injection
- With increasing numbers of lip augmentation procedures being carried out in clinical practice, it is important as an aesthetics practitioner to critically analyse the research available and ensure that you are providing the safest possible practice for your patients, while also minimising all potential risks.
CPD reflective questions
- What has research shown to be the ideal ratio of the upper and lower lip?
- What are the most important characteristics of a good dermal filler product?
- What are the main known causes of lip dermal filler migration?
- Are there any differences in risk of filler migration between the lip augmentation techniques of cannula versus needles?
- As an aesthetics practitioner, how would you go about minimising risks of lip dermal filler migration going forward now in your clinical practice?



